Introduction
Conducting polymers are a unique class of materials that exhibit properties of both metals and polymers. Poly(3,4-ethylenedioxythiophene) or PEDOT is one such conducting polymer that has gained significant attention in the last few decades due to its excellent properties and wide range of applications. In this article, we will explore the chemistry and properties of PEDOT that make it a promising material for numerous technological applications.
Discovery and Synthesis
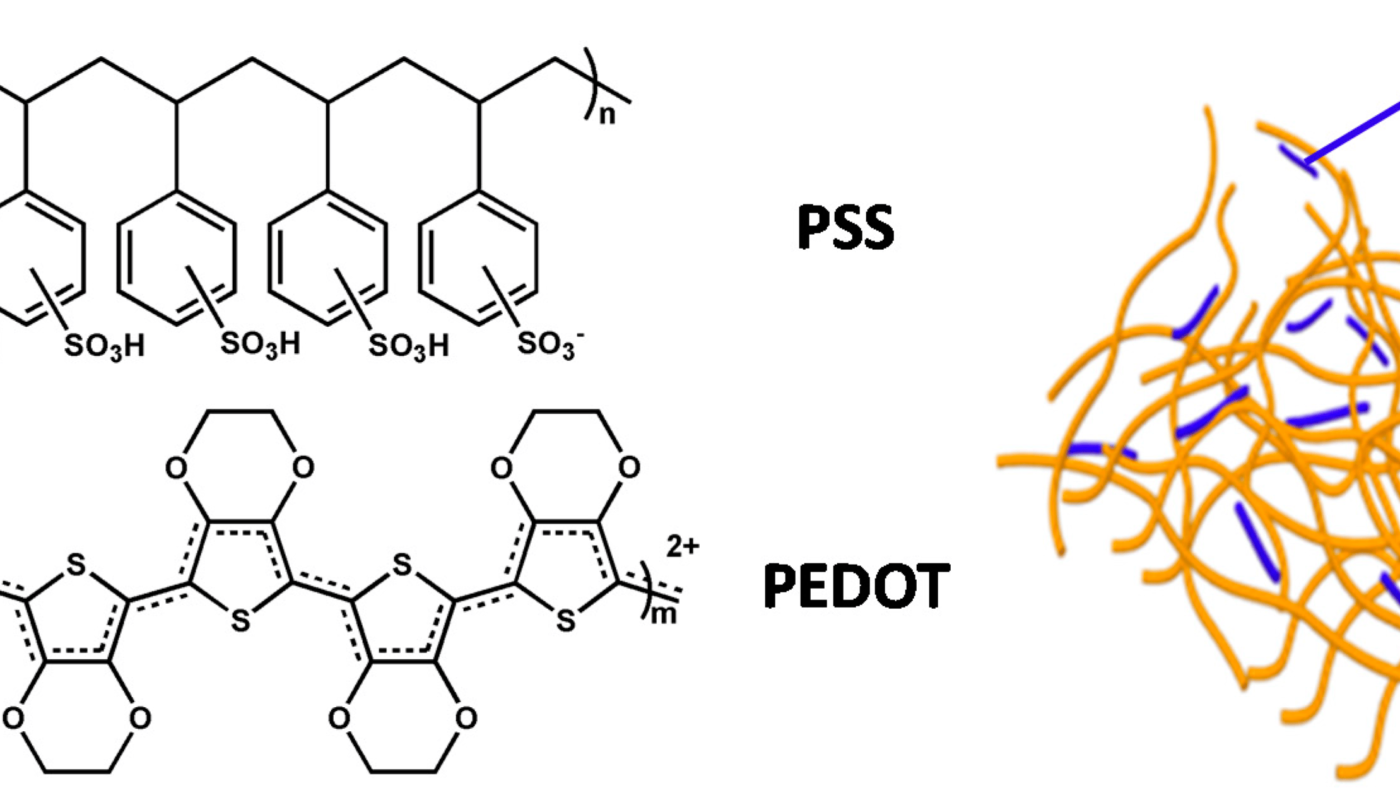
PEDOT was first synthesized and reported by researchers at Bayer AG in the 1980s. It is synthesized through the oxidative polymerization of its monomer, 3,4-ethylenedioxythiophene (EDOT) in the presence of an oxidizing agent. One of the most common oxidizing agents used is iron(III) chloride which helps dope the polymer during synthesis and impart conductivity. The polymerization proceeds through the formation of radical cations which then couple to form chains of connected thiophene rings.
Through careful control of parameters like monomer concentration, oxidant amount, polymerization temperature and time, PEDOT can be synthesized to have desired molecular weights, doping levels and dispersion properties. PEDOT has also been produced through electrolysis, electrochemical synthesis and chemical synthesis routes with varying properties. The polymer obtained is typically a blue-black powder or film with conductivity in the range of 1-1000 S/cm depending on synthesis and post-treatment methods.
Properties
Conductivity
As with other conducting polymers, PEDOT’s conductivity is attributed to the extensive conjugation and ability to be doped. Upon oxidation during synthesis, electrons are removed from the backbone generating holes or positive charges along the chains. These charges are mobile and allow charge transport, endowing PEDOT with conductivity. PEDOT can exhibit conductivity as high as 1000 S/cm, among the highest for conducting polymers.
Environmental Stability
PEDOT is known for its excellent environmental stability as the ethylenedioxy ring provides protection against oxidation and degradation. It maintains its conductivity even when exposed to ambient conditions for prolonged periods. This stability gives PEDOT an edge over other conducting polymers like polyaniline which tend to lose conductivity quickly due to oxidation.
Solubility and Processability
Unlike other less soluble conducting polymers, PEDOT can be produced in dispersions with particle sizes less than 50 nm, making it easily processable from aqueous media. It can be spin coated, inkjet printed, spray coated and electroplated depending on the application. This solubility and dispersion behavior has enabled PEDOT’s use in a diverse array of applications.
Applications of PEDOT
Biomedical Applications
Due to its biocompatibility, environmental stability and ability to be synthesized as conductive hydrogels and coatings, PEDOT is being widely researched for biomedical uses. These include neural electrodes for brain-machine interfaces, biosensors, tissue engineering scaffolds and substrates for growth of cells. PEDOT can emulate natural tissues better than inherently inorganic materials usually used.
Organic Electronics
As a key organic semiconductor and hole injection/transport material, PEDOT enjoys widespread applications in organic electronic devices. These include organic light emitting diodes, organic solar cells, organic field effect transistors and organic displays. The tunable properties and conductivity of PEDOT make it a versatile material for various organic device architectures.
Anti-corrosion Coatings
PEDOT coatings are being developed and commercialized for corrosion protection of metals especially in harsh, corrosive environments. The coatings are able to seal the metal surface and eliminate contact with corrosive electrolytes very effectively. PEDOT provides a barrier without compromising transparency, making it suitable for this application.
Touch Screens and Electrochromism
PEDOT dispersions are used frequently as transparent electrodes in touch screens and similar devices. It has also been applied to produce electrochromic devices which change color upon application of voltage due to the oxidation state change in PEDOT. This property has potential applications from smart windows to displays.
PEDOT stands out as a conducting polymer with excellent properties, versatility and environmental stability. Continuous research worldwide aims to enhance its processability, improve conductivity and utilize its biocompatibility. With new synthesis methods and nanostructured forms being developed, applications of PEDOT are bound to diversify and grow rapidly across various industries. It shows great potential to replace traditionally used materials and enable totally new technologies. PEDOT is thus a promising material for the future.
*Note:
1. Source: Coherent Market Insights, Public sources, Desk research
2. We have leveraged AI tools to mine information and compile it